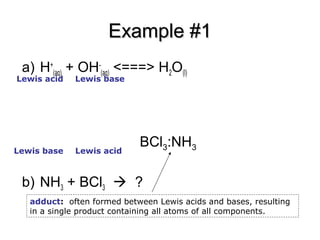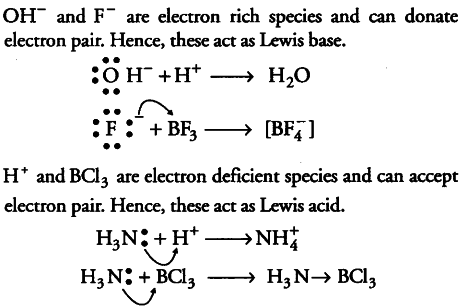
Classify the following species into Lewis acids and Lewis bases and show how these act as Lewis acid/base? - CBSE Class 11 Chemistry - Learn CBSE Forum

BCl3 lewis structure, molecular geometry, polar or nonpolar, hybridization, Bond angle in 2022 | Molecular geometry, Molecular, Vsepr theory

The Lewis acid character of boron trihalides decreases as: `B Br_(3) gt BCl_(3) gt BF_(3)`. - YouTube

Decide whether the given substance should be classified as a Lewis acid or a Lewis base.BCl3 (Hint; Draw the Lewis dot structure.) | Homework.Study.com
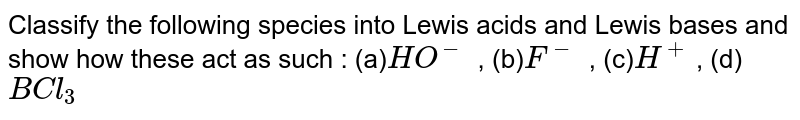
Classify the following species into Lewis acids and bases and show how these act as such: (i) BCl3 (ii) H^(+) (iii) F^(-) (iv) HO^(-)

From σ- to π-Electrophilic Lewis Acids. Application to Selective Organic Transformations | The Journal of Organic Chemistry