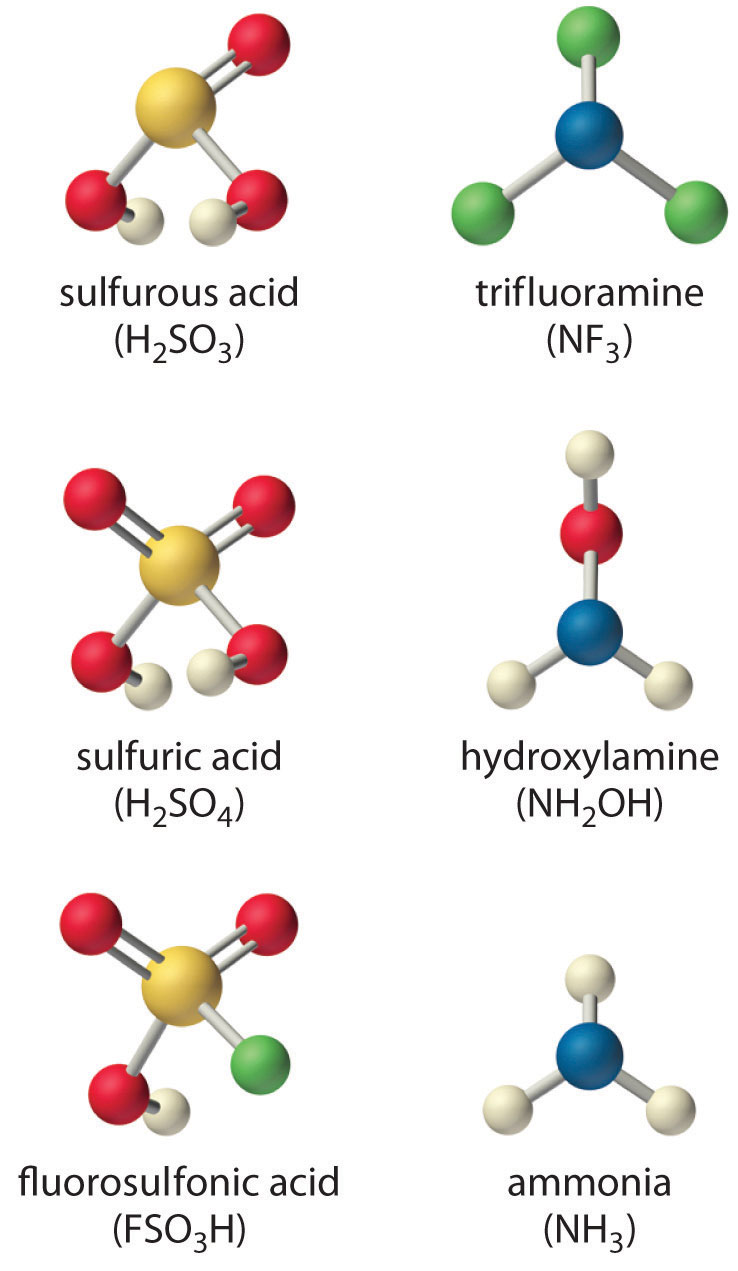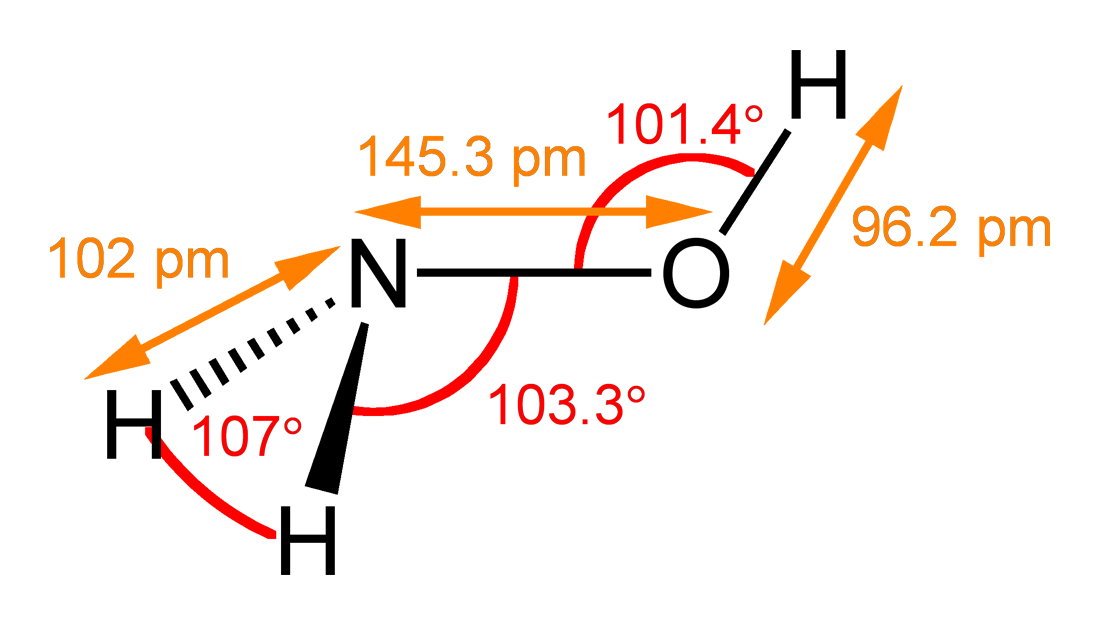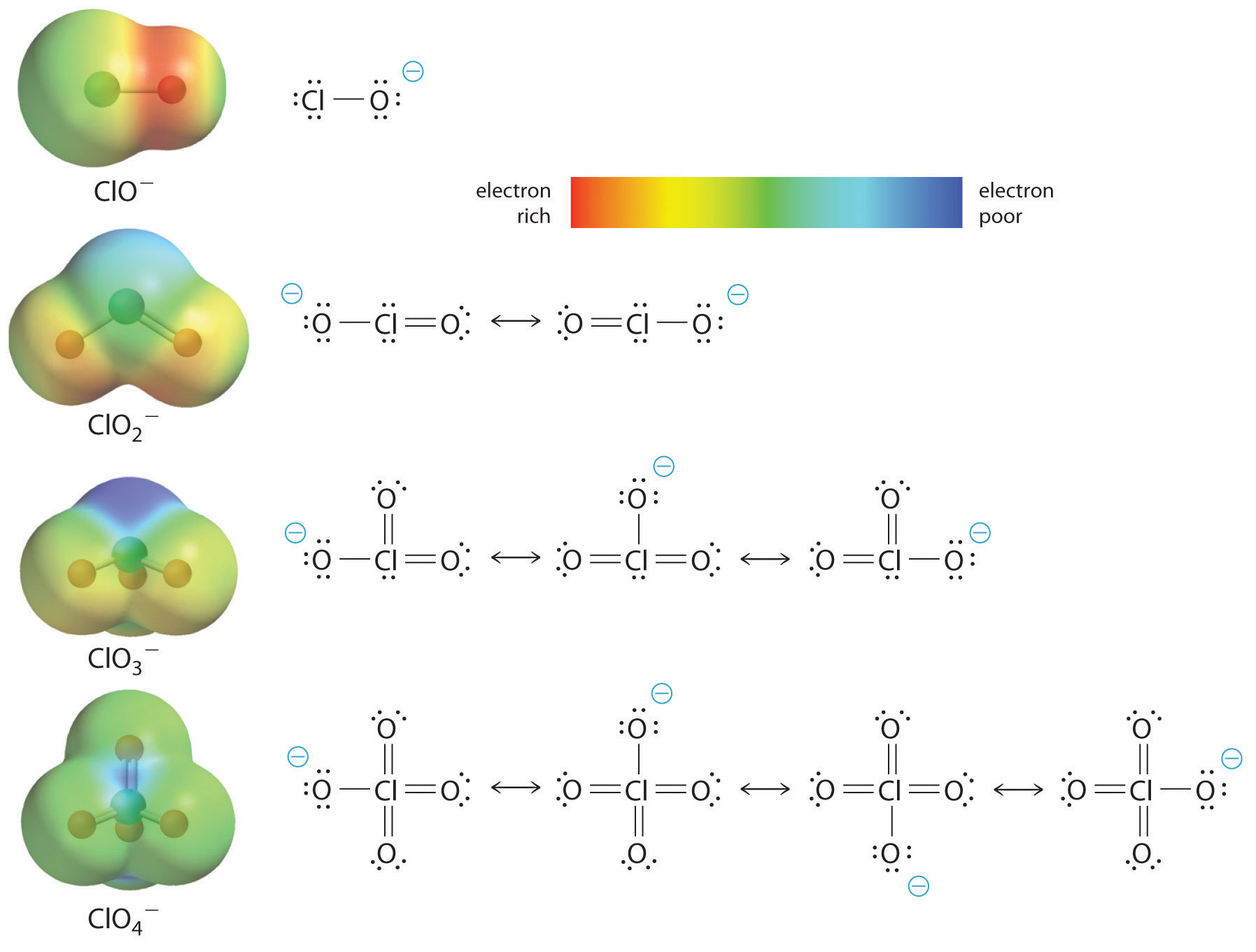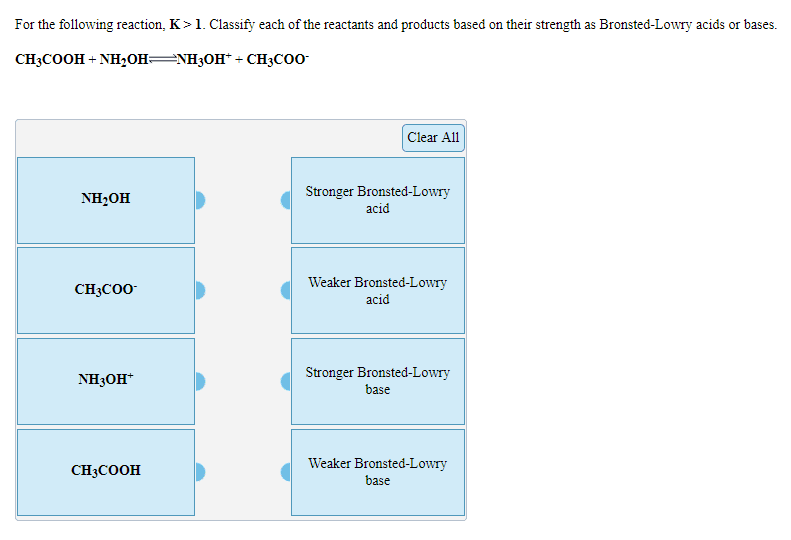
OneClass: For the following reaction, K>1. Classify each of the reactants and products based on th...
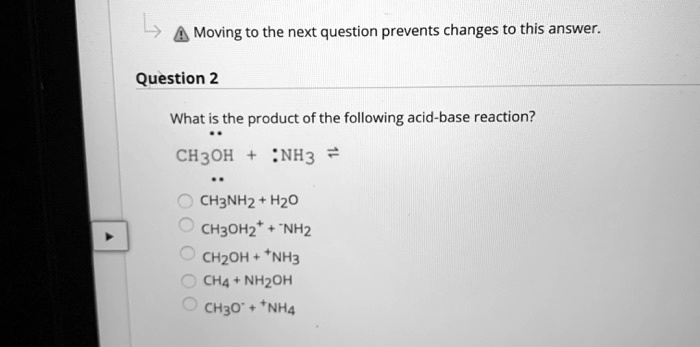
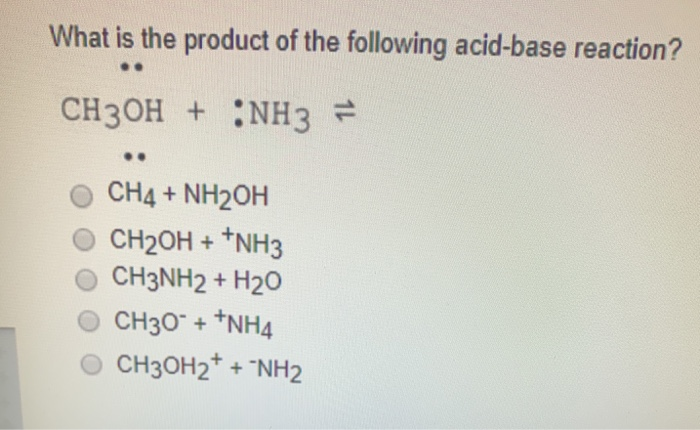
SOLVED: Moving to the next question prevents changes to this answer: Question 2 What is the product of the following acid-base reaction? CH3OH NH3 CH3NH2 Hzo CH30Hz* NH2 CH2OH "NH3 CHA NH2OH
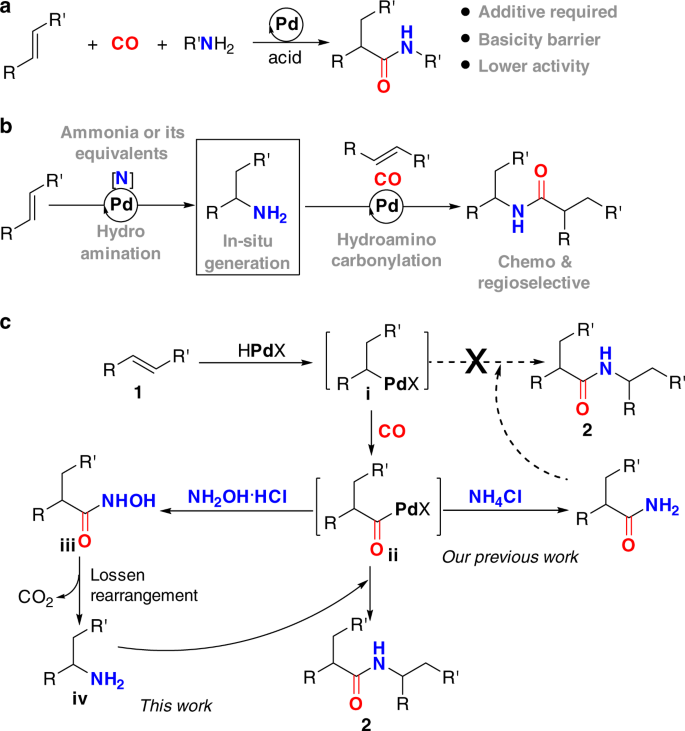
Palladium-catalyzed relay hydroaminocarbonylation of alkenes with hydroxylamine hydrochloride as an ammonia equivalent | Communications Chemistry

Chapter 15: Acids and Bases Bronsted Lowry Acids HCl H + (aq) + Cl - (aq) HNO 3 H + (aq) + NO 3 - (aq) HC 2 H 3 O 2 H + (aq) + C 2 H 3 O 2 - (aq) - ppt download

Palladium-catalyzed relay hydroaminocarbonylation of alkenes with hydroxylamine hydrochloride as an ammonia equivalent | Communications Chemistry

OneClass: Label the Bronsted-Lowry acid and base as well as the conjugate acid and conjugate base for...
Synthesis of Hydroxamic Acids by Activation of Carboxylic Acids with N,N′-Carbonyldiimidazole: Exploring the Efficiency of the

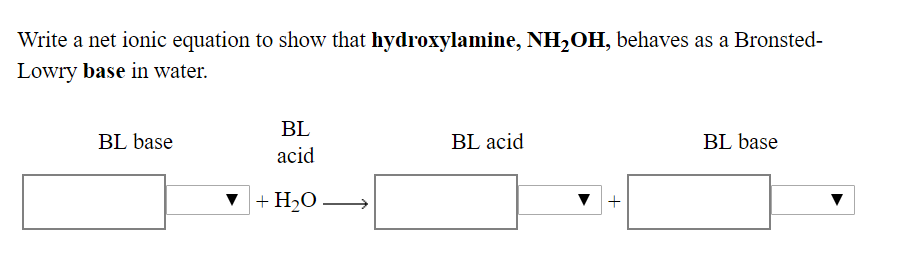
SOLVED: 1. Write the name of all strong acids and bases. 2. What is the difference between a strong and weak acid and bases? 3. Hydroxylamine (NH2OH) is a weak base with

Reagents and conditions: (a) CCl3CH(OH)2, NH2OH. HCl, 45min heating;... | Download Scientific Diagram

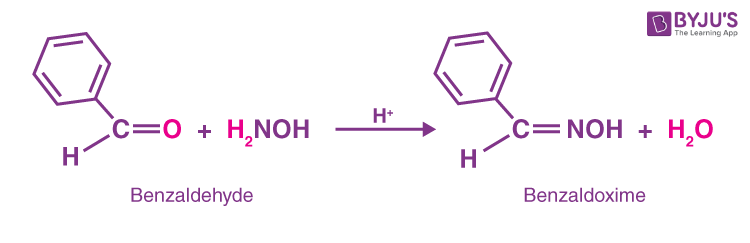
Photoorganocatalytic One‐Pot Synthesis of Hydroxamic Acids from Aldehydes - Papadopoulos - 2016 - Chemistry – A European Journal - Wiley Online Library